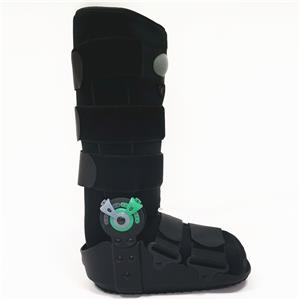
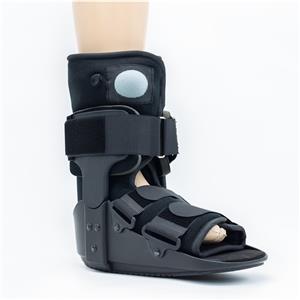
From Zero to One: Our 180-Day Hard-Fought Battle to Perfect the Carbon Fiber AFO
The Crack That Started It AllEarly spring 2025. Our production workshop fell quiet, save for the hum of idle equipment.
In our hands was the 37th failed trial sample of our carbon fiber AFO.
A thin delamination crack along its curved corner stood like a glaring chasm, between us and our mission: to build a medical-grade carbon fiber AFO accessible to patients across the globe. ---The Gap We Couldn’t IgnoreFor over a decade, we’ve been deeply rooted in the orthopedic brace industry, and we’ve seen the industry’s unspoken frustrations firsthand.
We watched stroke survivors with foot drop sweat through their clothes after just a few steps, weighed down by bulky plastic AFOs.
We saw children with hemiplegia miss their critical recovery window, locked out by imported carbon fiber braces that cost tens of thousands of yuan.
Overseas brands held a tight stranglehold on core technology and pricing power. Most domestic alternatives were non-standard industrial-grade carbon fiber parts that failed to meet medical requirements.
*If they can do it, why can't we?*
With that question, we threw ourselves headlong into an uphill battle that would consume the next 180 days.
---The Gap We Couldn’t IgnoreFor over a decade, we’ve been deeply rooted in the orthopedic brace industry, and we’ve seen the industry’s unspoken frustrations firsthand.
We watched stroke survivors with foot drop sweat through their clothes after just a few steps, weighed down by bulky plastic AFOs.
We saw children with hemiplegia miss their critical recovery window, locked out by imported carbon fiber braces that cost tens of thousands of yuan.
Overseas brands held a tight stranglehold on core technology and pricing power. Most domestic alternatives were non-standard industrial-grade carbon fiber parts that failed to meet medical requirements.
*If they can do it, why can't we?*
With that question, we threw ourselves headlong into an uphill battle that would consume the next 180 days. ---The First Hurdle: Cracking the Medical-Grade FormulaThe core of a carbon fiber AFO lies in a near-impossible balance: rigidity and flexibility.
It must be stiff enough to stabilize a weakened ankle and correct foot drop with every step. It must also deliver just the right resilient rebound to restore a natural, smooth gait.
We sourced materials from dozens of manufacturers nationwide, but our early trials hit a brick wall.
Samples rigid enough for clinical support shattered like glass in drop tests. Samples with enough flexibility lacked the stability to correct abnormal gait, falling completely short of clinical standards.
Our R&D team locked themselves in the lab, refining every single parameter down to the millimeter.
We tweaked the weave density of carbon fiber filaments, adjusted the ratio of medical-grade resin matrix down to a single percentage point, and ran full tests on every formula one by one.
We conducted hundreds of rounds of testing on four core metrics alone: tensile strength, bending performance, fatigue resistance, and biocompatibility. Discarded samples piled up half the lab.
At last, we landed on our exclusive formula: one that perfectly balanced uncompromising medical safety, clinical-grade rigidity, and tailored flexibility.
---The Make-or-Break Challenge: Mastering the Molding ProcessWe barely had time to catch our breath before an even harder challenge hit: perfecting the molding process.
An AFO is a complex curved component, precision-engineered to fit the natural physiological curve of the human ankle. It needs an ultra-thin wall thickness of less than 1mm to fit inside everyday footwear — an extreme test for carbon fiber integrated molding technology.
Our first batch of 100 trial-molded samples, made with traditional autoclave molding, had a yield rate of less than 5%. Air bubbles, delamination, and incomplete filling at the curved corners plagued every run.
We revised the mold time and time again, adjusting every single variable: demolding angles, carbon fiber layup design, hot pressing temperature, and pressure holding time.
Our lead process engineer lived and slept in the workshop for 42 consecutive days, sleeping only 3 to 4 hours a night, just to monitor every second of the molding cycle and catch the tiniest one-in-ten-thousand error.
We scrapped 3 complete molding solutions and revised the mold 17 times.
Finally, we lifted the yield rate from 5% to over 98%. For the first time, we saw a light at the end of the tunnel.
---The First Hurdle: Cracking the Medical-Grade FormulaThe core of a carbon fiber AFO lies in a near-impossible balance: rigidity and flexibility.
It must be stiff enough to stabilize a weakened ankle and correct foot drop with every step. It must also deliver just the right resilient rebound to restore a natural, smooth gait.
We sourced materials from dozens of manufacturers nationwide, but our early trials hit a brick wall.
Samples rigid enough for clinical support shattered like glass in drop tests. Samples with enough flexibility lacked the stability to correct abnormal gait, falling completely short of clinical standards.
Our R&D team locked themselves in the lab, refining every single parameter down to the millimeter.
We tweaked the weave density of carbon fiber filaments, adjusted the ratio of medical-grade resin matrix down to a single percentage point, and ran full tests on every formula one by one.
We conducted hundreds of rounds of testing on four core metrics alone: tensile strength, bending performance, fatigue resistance, and biocompatibility. Discarded samples piled up half the lab.
At last, we landed on our exclusive formula: one that perfectly balanced uncompromising medical safety, clinical-grade rigidity, and tailored flexibility.
---The Make-or-Break Challenge: Mastering the Molding ProcessWe barely had time to catch our breath before an even harder challenge hit: perfecting the molding process.
An AFO is a complex curved component, precision-engineered to fit the natural physiological curve of the human ankle. It needs an ultra-thin wall thickness of less than 1mm to fit inside everyday footwear — an extreme test for carbon fiber integrated molding technology.
Our first batch of 100 trial-molded samples, made with traditional autoclave molding, had a yield rate of less than 5%. Air bubbles, delamination, and incomplete filling at the curved corners plagued every run.
We revised the mold time and time again, adjusting every single variable: demolding angles, carbon fiber layup design, hot pressing temperature, and pressure holding time.
Our lead process engineer lived and slept in the workshop for 42 consecutive days, sleeping only 3 to 4 hours a night, just to monitor every second of the molding cycle and catch the tiniest one-in-ten-thousand error.
We scrapped 3 complete molding solutions and revised the mold 17 times.
Finally, we lifted the yield rate from 5% to over 98%. For the first time, we saw a light at the end of the tunnel. ---The Final Test: Holding the Line on Medical StandardsThe last life-or-death hurdle was the non-negotiable bottom line of medical compliance.
To reach patients worldwide, we had to pass CE and FDA certification. The fatigue test alone required simulating a normal human gait and completing 100,000 loading cycles without deformation or fracture.
Our 52nd optimized sample failed at 32,000 cycles. A tiny crack appeared at the ankle’s stress concentration point.
The entire team fell silent.
Someone spoke up: “Can’t we lower the standards a little? It’s still good enough for daily use.”
But we knew better than anyone: for patients in rehabilitation, even this slight “shortfall” could lead to a fall, or a complete interruption of their recovery process.
We refused to cut corners.
We tore the design apart, used finite element analysis (FEA) to map the force trajectory of every single step, optimized the layup direction, and added an invisible reinforcement design at the stress concentration point.
After more than a dozen rounds of repeated tests, our sample finally passed the full 100,000-cycle fatigue test with flying colors. Every medical safety check was fully cleared.
---The Final Test: Holding the Line on Medical StandardsThe last life-or-death hurdle was the non-negotiable bottom line of medical compliance.
To reach patients worldwide, we had to pass CE and FDA certification. The fatigue test alone required simulating a normal human gait and completing 100,000 loading cycles without deformation or fracture.
Our 52nd optimized sample failed at 32,000 cycles. A tiny crack appeared at the ankle’s stress concentration point.
The entire team fell silent.
Someone spoke up: “Can’t we lower the standards a little? It’s still good enough for daily use.”
But we knew better than anyone: for patients in rehabilitation, even this slight “shortfall” could lead to a fall, or a complete interruption of their recovery process.
We refused to cut corners.
We tore the design apart, used finite element analysis (FEA) to map the force trajectory of every single step, optimized the layup direction, and added an invisible reinforcement design at the stress concentration point.
After more than a dozen rounds of repeated tests, our sample finally passed the full 100,000-cycle fatigue test with flying colors. Every medical safety check was fully cleared.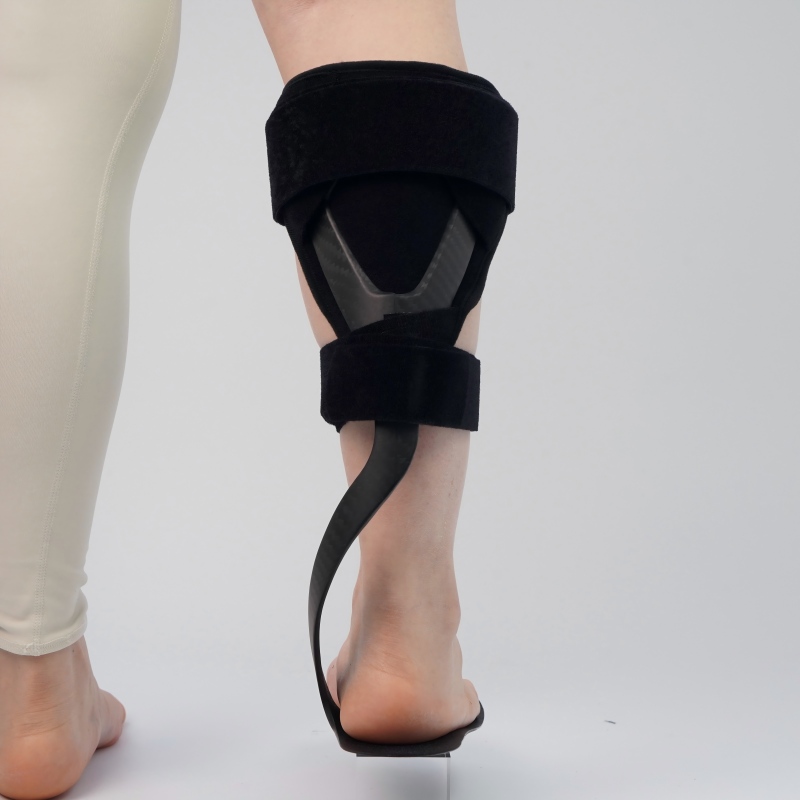 ---
---
The Finish Line: More Than Just a Product180 days of relentless work. 37 full trial runs. 17 mold revisions. Hundreds of lab tests. The first mass-produced batch of our carbon fiber AFO rolled smoothly off the production line. We held it up next to the leading imported benchmark — ours was lighter, stronger, and cost less than a third of the price. Today, our carbon fiber AFOs are used in rehabilitation facilities and hospitals in more than 20 countries around the world. We once received an email from a rehabilitation therapist in the Netherlands. She told us about a patient who had been bedridden for six months after a stroke. With our AFO, he walked to the window on his own for the first time, and got to see the spring tulips in full bloom. In that moment, we finally understood: every all-nighter we pulled, every ruined sample, every time we gritted our teeth and refused to give up, had found its most precious meaning. The road from zero to one was arduous. But at the end of it isn’t just a product. It’s the chance for more patients to take their first step back into life.