MDR CE, FDA & ISO 13485 Certified Orthopedic Rehabilitation Braces OEM/ODM Manufacturer | On-Site Meetings in Europe: May 18 – June 10, 2026
As a professional OEM/ODM orthopedic braces manufacturer with over 12 years of industry experience, Xiamen Huakang Orthopedic Co., Ltd. is thrilled to announce our official Europe roadshow from May 18 to June 10, 2026. We are heading to Europe to hold face-to-face on-site meetings with local orthopedic device brands, distributors, and medical partners, bringing our full range of MDR CE certified orthopedic rehabilitation braces, end-to-end custom orthopedic OEM/ODM solutions, and exclusive European market cooperation policies directly to you.
For years, we have been a trusted long-term partner for European clients in the orthopedic and rehabilitation medical device field, specializing in custom production of compliant, high-quality orthopedic braces that fully meet EU market access standards. This on-site roadshow is our commitment to European partners: we want to connect with you in person, let you experience our products first-hand, and build deeper, more transparent OEM/ODM cooperation for your orthopedic device business.
Why Choose Huakang Ortho as Your Orthopedic Braces OEM/ODM Partner?
1. Full Regulatory Compliance for the European Orthopedic Device Market
The most critical threshold for the European orthopedic device market is regulatory compliance, and we have fully prepared for you:
We hold MDR CE certification for orthopedic medical devices, fully meeting the latest EU medical device regulations, which is the core access qualification for your products to enter the European market
We have obtained FDA certification and ISO 13485 medical device quality management system certification, with a complete quality control system that meets global mainstream medical device market standards
All our OEM/ODM orthopedic braces can provide full compliance document support, helping your brand complete product registration and market entry in the EU efficiently
2. Full Range of Customizable OEM/ODM Orthopedic Braces & Rehabilitation Devices
Our OEM/ODM orthopedic product portfolio covers full-body orthopedic and rehabilitation solutions, with all hot-selling products verified by the European market, and can be fully customized according to your brand’s needs. Our customizable product line includes:
OEM OA knee braces, ROM dual OA knee braces, offloading osteoarthritis knee braces, pediatric adjustable quick-locking ROM knee braces
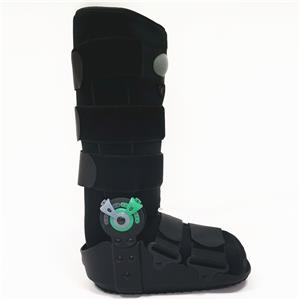
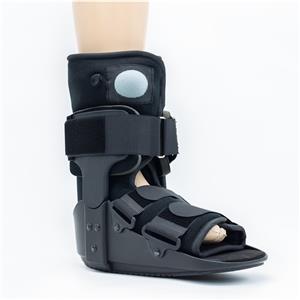
OEM walker boots, tall poly walking boots with air liner, short poly walker boots with airbag and rocker bottom, tall pneumatic ROM walker boot braces
OEM ankle braces, ankle foot orthosis (AFO), night splints, neoprene calf sport braces
OEM back supports, lower back braces with dual adjustable straps and back plate, lumbar support systems
OEM cervical collars, soft collars with semi-hard thermoplastic panel, CTO (Cervical Vertebrae-Thoracic Orthosis)
OEM elbow braces, arm slings, upper limb orthopedic rehabilitation braces, and other full-body protective and orthopedic equipment
3. End-to-End OEM/ODM Capabilities for Orthopedic Medical Devices
Custom product design and R&D: Our professional R&D team can develop exclusive orthopedic brace products according to your brand positioning, market demand and functional requirements
Private label orthopedic braces full customization: Support brand logo printing, exclusive packaging design, product specification customization, to build your brand’s exclusive product image
Flexible MOQ setting: We provide friendly minimum order quantity for European brands of all sizes, whether you are a start-up brand or a large-scale distributor, we can match the most suitable cooperation plan
Full technical and after-sales support: From pre-production prototype confirmation to after-sales market support, we provide full-process tracking service for your OEM/ODM orthopedic projects
4. Strict Quality Control & Stable Mass Production for European Partners
Founded in 2014, we have built a complete modern production system and strict quality control system, with rich experience in bulk shipment to the European market. Our production capacity and quality control system can fully meet the stable supply needs of European partners, ensuring that every batch of OEM/ODM orthopedic braces delivered to you meets the highest medical device quality standards.

Meet Us Face-to-Face in Europe | May 18 – June 10, 2026
During our Europe roadshow from May 18 to June 10, 2026, we welcome all European orthopedic device brands, distributors, and medical partners to book a 1-on-1 on-site meeting with our professional sales and R&D team.
What you can get from the face-to-face meeting:
On-site sample viewing and hands-on testing of our full range of MDR CE certified orthopedic braces, to intuitively experience the product quality, material and functional design
In-depth discussion of your custom OEM/ODM orthopedic project requirements directly with our core team, to confirm product details, customization plans and production cycles on the spot
Exclusive cooperation terms and preferential policies for European orthopedic partners, only for customers who book on-site meetings during the roadshow
Full details of our production capacity, quality control system, and successful European market cooperation cases, to build a transparent and trustworthy long-term cooperation foundation
Book Your 1-on-1 Meeting Now
📅 Roadshow Time: May 18 – June 10, 2026
🌐 Official Website: https://www.orthotic-braces.com/ (View our full product line and OEM/ODM capabilities)
📩 Booking Email: sales6@reabletrading.net
📱 WhatsApp/WeChat: +86-18030275537